Alpha Sophia for Real-World Data & Evidence (RWD & RWE)
Challenge


In the current healthcare environment, clinical trials are only the beginning of a product's story. To truly understand how a drug or medical device performs, organizations must bridge the gap between controlled environments and the "messy" reality of clinical practice. However, accessing and analyzing Real-World Data (RWD) and generating Real-World Evidence (RWE) is often hampered by fragmentation. Data is scattered across disparate systems, making it difficult to measure the true effectiveness, safety, and economic value of treatments in diverse patient populations.
Several structural hurdles frequently limit the impact of RWE initiatives:
- The "Clinical Reality" Gap:Many teams rely on small, internal data sets that don't reflect the broader market. Without a unified view of all-payor claims data and physician-level activity, organizations struggle to see how their products are actually being used by clinicians on the front lines.
- Fragmented Longitudinal Insights:Understanding the patient journey—from first symptom to long-term follow-up—is nearly impossible when data is siloed. This fragmentation prevents teams from demonstrating lab value in value-based care models and proving the long-term ROI of specialized treatments.
- Lagging Intent Signals:Relying on data that is years old makes it impossible to react to emerging clinical trends. To be effective, RWE must be built on a foundation of real-world HCP data trends that reflect current practice patterns, not historical averages.
- Difficulty in Proving Value to Payors:In a landscape defined by high transparency and pricing scrutiny, simply saying a product works isn't enough. Organizations need hard evidence of clinical outcomes and cost-efficiency to justify reimbursement and secure a spot on the formulary.
Solution
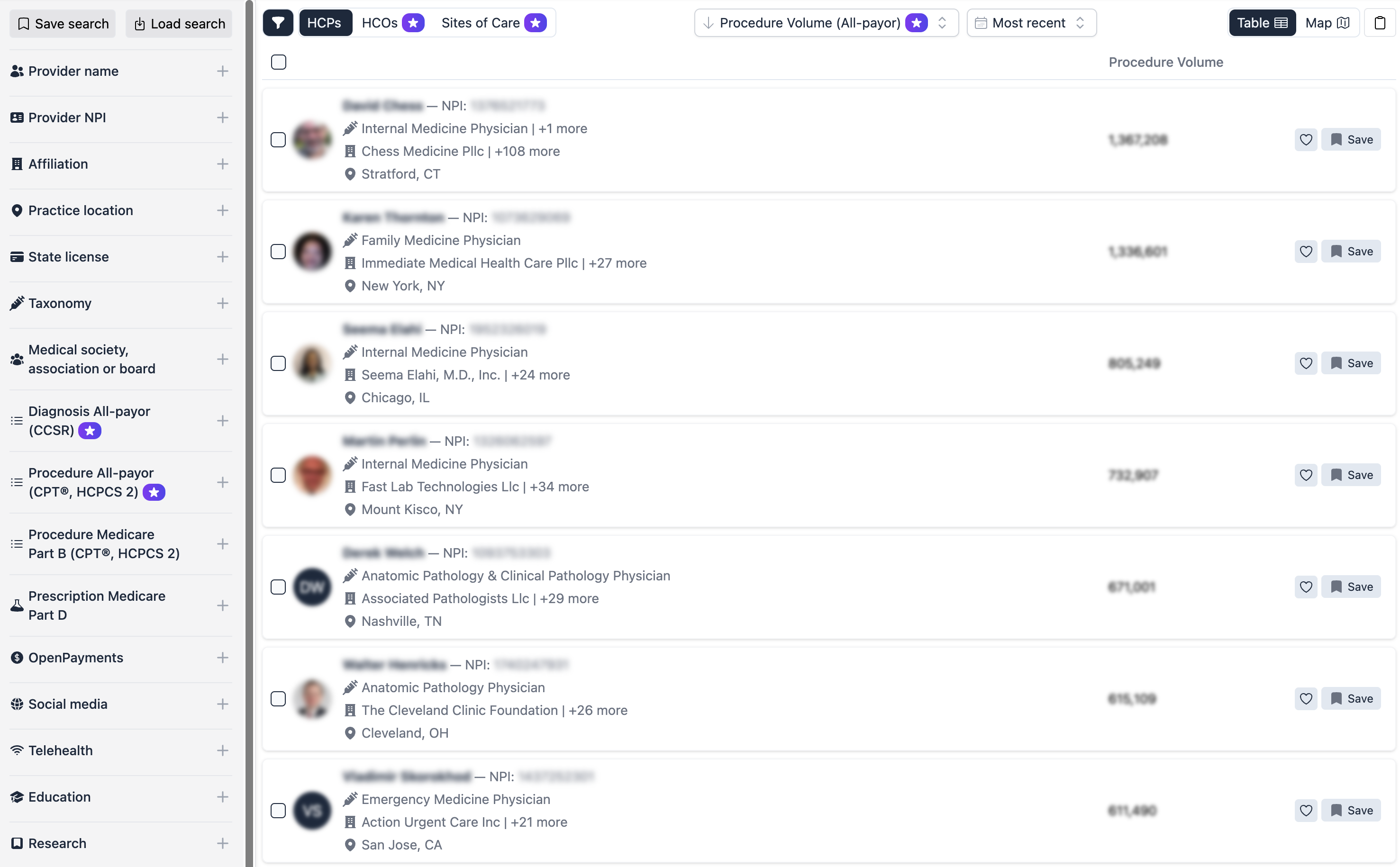

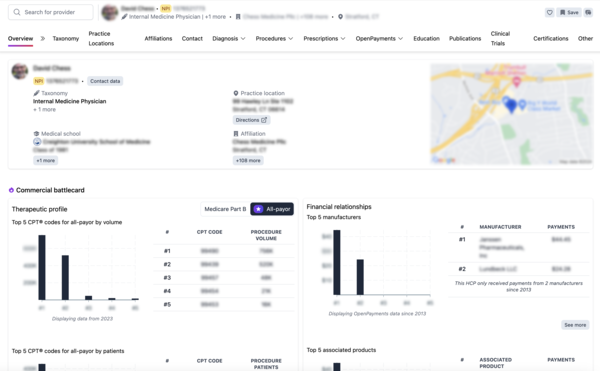
Alpha Sophia provides a unified commercial intelligence platform that consolidates high-fidelity Real-World Data into actionable insights. By offering detailed profiles of HCPs and HCOs—including CPT/HCPCS procedure volumes, ICD-10 diagnosis patterns, and professional affiliations—Alpha Sophia allows organizations to measure the true footprint of their products across the entire healthcare ecosystem.
With Alpha Sophia, teams can:
- Benchmark Real-World Performance:Use all-payer claims data to see how your treatment adoption compares to the standard of care. This allows you to identify which provider segments are achieving the best results and why.
- Map the Complete Patient Journey:Filter by diagnosis and procedure clusters to understand the longitudinal pathway of patients. This is a core part of Alpha Sophia for Real-World Data and Evidence, helping you identify exactly where treatments are most effective.
- Analyze Telehealth and Digital Adoption:Assess how virtual care and digital monitoring impact treatment adherence and outcomes by tracking the 2026 telehealth landscape and its role in modern care delivery.
- Refine Market Strategy with Hard Evidence:Use Open Payments and research engagement data to contextualize your clinical evidence. By identifying "Digital Opinion Leaders" who are already documenting real-world successes, you can amplify your RWE through trusted clinical voices.
Example
A medical device company specializing in minimally invasive spine surgery can use Alpha Sophia to track the real-world utilization of their implant versus traditional fusion. By analyzing the longitudinal billing patterns of surgeons, they can prove that patients treated with their device have fewer 90-day re-admissions and require significantly less post-operative physical therapy. This RWE is then used to negotiate preferred status with major health insurers, proving that the device lowers the total cost of care while improving patient recovery times.